Top health and wellness news from New Mexico
Provided by AGPBriaCell Receives FDA Clearance to Initiate Bria-BRES+™ Clinical Study in Breast Cancer
- FDA clearance of Investigational New Drug (IND) application opens path to commence Phase 1/2a clinical study for Bria-BRES+ in metastatic breast cancer
- Bria-BRES+ is BriaCell’s next generation personalized immunotherapy for breast cancer, and features additional immune activating components designed to enhance clinical efficacy
-
BriaCell has prepared clinical supplies of Bria-BRES+ and plans to initiate a Phase 1/2a clinical study in metastatic breast cancer in the coming months
PHILADELPHIA and VANCOUVER, British Columbia, May 06, 2026 (GLOBE NEWSWIRE) -- BriaCell Therapeutics Corp. (Nasdaq: BCTX, BCTXL) (TSX: BCT) (“BriaCell” or the “Company”), a clinical-stage biotechnology company developing novel immunotherapies to transform cancer care, is pleased to report it has received FDA clearance to initiate clinical evaluation of Bria-BRES+, its next generation, personalized, off-the-shelf, cell-based immunotherapy for metastatic breast cancer.
“We are honored to announce FDA clearance of the first IND for our next generation personalized immunotherapy, Bria-BRES+,” stated Dr. William V. Williams, BriaCell’s President & CEO. “The unique design of Bria-BRES+ offers the potential for a favorable safety profile and meaningful therapeutic benefit in metastatic breast cancer. We look forward to advancing Bria-BRES+ into the clinic as we seek to bring new hope to these patients who have few to no effective treatment options.”
As reported in BriaCell’s recent AACR preclinical poster presentation, Bria-BRES+ demonstrated activation of both adaptive and innate immunity including activation of naïve (resting) T-cells, dendritic cells and natural killer (NK) cells. BriaCell believes this multipronged immune activation may enhance clinical efficacy and help prevent immune escape in patients with metastatic breast cancer.
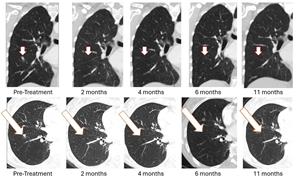
BriaCell’s Bria-BRES+ builds on its Bria-OTS™ breast cancer clinical program, where the first patient dosed experienced the sustained complete resolution of a lung metastasis. This 78-year-old woman with advanced metastatic breast cancer and multiple prior treatment failures achieved complete (100%) resolution of a lung metastasis following four doses of Bria-OTS single agent therapy. The complete response of the lesion, initially observed at 2 months, was subsequently confirmed at 4 months, 6 months, and at 11 months. The patient received 17 cycles of Bria-OTS, completed 12 months of the study, and remains in survival follow-up.
Figure 1: As previously reported in January 2026, treatment with Bria-OTS monotherapy resulted in 100% resolution of tumor in the right lung of the metastatic breast cancer (MBC) patient following 2 months of therapy and confirmed at 4, 6, and 11 months of therapy1 (axial and coronal views)
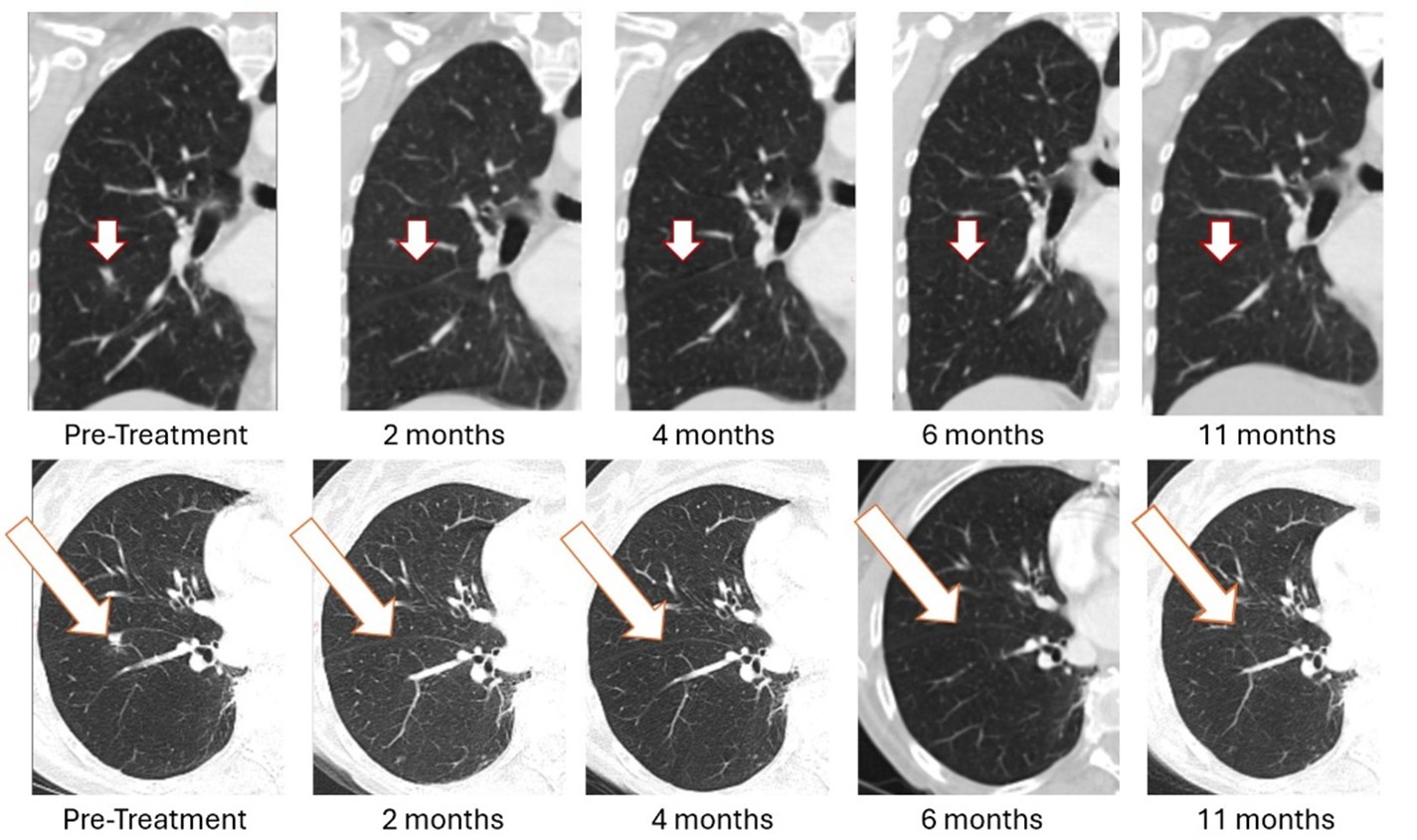
1 Note that the other white dots in the lungs are blood vessels.
About BriaCell Therapeutics Corp.
BriaCell is an immuno-oncology-focused biotechnology company developing targeted and effective approaches for the management of cancer. More information is available at https://briacell.com/.
Safe Harbor
This press release contains “forward-looking statements” that are subject to substantial risks and uncertainties. All statements, other than statements of historical fact, contained in this press release are forward-looking statements. Forward-looking statements contained in this press release may be identified by the use of words such as “anticipate,” “believe,” “contemplate,” “could,” “estimate,” “expect,” “intend,” “seek,” “may,” “might,” “plan,” “potential,” “predict,” “project,” “target,” “aim,” “should,” “will,” “would,” or the negative of these words or other similar expressions, although not all forward-looking statements contain these words. Forward-looking statements, including those about the FDA clearance of the IND application and the planned initiation of a Phase 1/2a clinical study of Bria‑BRES+, and expectations regarding the potential clinical benefits of the Bria‑OTS+ platform, are based on BriaCell’s current expectations and are subject to inherent uncertainties, risks, and assumptions that are difficult to predict. Further, certain forward-looking statements are based on assumptions as to future events that may not prove to be accurate. These and other risks and uncertainties are described more fully under the heading “Risks and Uncertainties” in the Company's most recent Management’s Discussion and Analysis, under the heading "Risk Factors" in the Company's most recent Annual Information Form, and under “Risks and Uncertainties” in the Company's other filings with the Canadian securities regulatory authorities and the U.S. Securities and Exchange Commission, all of which are available under the Company's profiles on SEDAR+ at www.sedarplus.ca and on EDGAR at www.sec.gov. Forward-looking statements contained in this announcement are made as of this date, and BriaCell Therapeutics Corp. undertakes no duty to update such information except as required under applicable law.
Neither the Toronto Stock Exchange nor its Regulation Services Provider (as that term is defined in the policies of the Toronto Stock Exchange) accepts responsibility for the adequacy or accuracy of this release.
Contact Information
Company Contact:
William V. Williams, MD
President & CEO
1-888-485-6340
info@briacell.com
Investor Relations Contact:
investors@briacell.com
A photo accompanying this announcement is available at https://www.globenewswire.com/NewsRoom/AttachmentNg/cf1af19a-fd92-49b4-8e39-fb9b1c4191a7

Legal Disclaimer:
EIN Presswire provides this news content "as is" without warranty of any kind. We do not accept any responsibility or liability for the accuracy, content, images, videos, licenses, completeness, legality, or reliability of the information contained in this article. If you have any complaints or copyright issues related to this article, kindly contact the author above.